Managing patient-monitoring equipment typically used for measuring patients’ heart rate, blood pressure and other vital signs goes far beyond simple inventory control, to include the need to ensure inspection, calibration, self-test results and safety upgrades, while minimizing down time. Adhesive paper labels applied to equipment and used to record service data are no longer a reasonable option, due to the large amount of information required, as well as the tendency for such labels to become damaged over time. With the technology advancing quickly, patient-monitoring equipment is often capable of being upgraded via software.
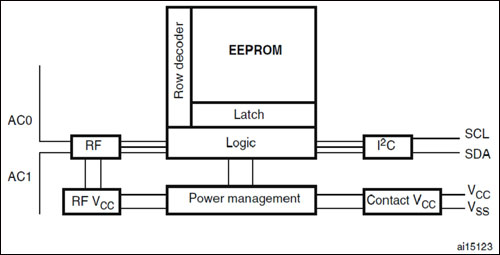
A dual-interface, RFID-based EEPROM (electrically erasable programmable read-only memory) solution is dynamic, and can record internal parameters for later readout, as well as write new data into the system, such as calibration constants and inspection information, without the need for additional connectors. A dual-interface RFID tag can be connected to electronic patient-monitoring equipment via an I²C port, and the tag’s memory can be read and written to using that port while the equipment is in operation. Reading can also be accomplished regardless of power to the equipment, via a conventional RFID reader compliant with the ISO 15693 standard for 13.56 MHz RFID tags. The ability to have this data current, secured yet readily accessible when needed completes the technology chain.
|
|
Applications for a dual-interface passive RFID system include access to maintenance requirements and records, verification of approved accessories, sensors and related components, and counterfeit goods rejection, as well as ensuring that one-time use items are discarded and not reused, and the addition of new approved devices as available. The advantage of a dual-interface RFID solution opens a designer’s options by allowing data to be managed from the monitoring equipment during operation or standby, while enabling the reading and writing of data via RFID readers, even if the equipment is powered down.
Monitoring System Categories
Patient-monitoring systems typically fall into three broad categories: bedside, portable and body-worn.
Bedside monitors play a significant role in providing ever-increasing portions of the information required by health-care professionals. These devices are often assigned to specific areas within a medical facility, such as critical and intensive-care units, and most are now capable of connecting with a central monitoring system on a hospital network, on which data can be exchanged through a facilities network.
Portable monitors are a bit more challenging, as they seem to have the ability to walk off and become lost. Although detecting location is not part of this discussion, knowing what happened to equipment can be a big help in addressing compliance issues and validating ownership.
Body-worn monitors are not new, but the types of measurements and amounts of data involved are increasing rapidly with each successive generation. This is an application in which a dual-interface RFID solution could greatly enhance the monitors’ utility and longevity, by providing a gateway to the inner workings without a cumbersome connector.
Within the body-worn categories, there are several subcategories:
• Mobile/wearable personal monitoring (MPM): Devices that monitor in real time, as well as store and forward data or send alerts regarding biometric activity for chronic diseases.
• Mobile aggregators: Smartphone-like devices with or without add-on sensors having the ability to report patient status using wireless technology.
• Wearable health gadgets: Devices that employ sensors worn on the wrist, arm or chest, or embedded in fabric in shoes or shirts, to measure heart rate, respiration, pace and more, for fitness, wellness and preventive care.
• Remote patient management (RPM): Specialized monitoring devices with sensors specific to a patient’s particular needs. Such systems can be custom-fitted with sensors to report everything from heart rate to whether a patient is standing or lying down.
For all types of patient monitors, whether bedside, portable or body-worn, the challenges remain: How do you keep equipment up to date with current software, calibration and maintenance records? And how do you know when a fault has occurred?
Benefits of Managing System Data
A simple failure can make the difference in regard to patient outcome. It should come as no surprise that the failure of a monitoring device’s battery backup is high on the list of issues plaguing the industry. System self-test alerts can go unreported, and can later reoccur at the most inopportune moments. For bedside monitors, this can be reported through a central monitoring capability, and corrective action can be performed, thus averting a serious problem.
Portable and body-worn monitors present a more challenging array of issues. One is that these segments are the fastest growing, and interoperability standards are only recently becoming a clear focus. A recent example is the Continua Health Alliance, which has selected four main interfaces for interoperability: USB, Bluetooth, Bluetooth Low Energy (BTLE) and ZigBee. The common thread here is that the monitors need to be powered up and operating (that is, in service) for these interfaces to be reporting, thus indicating they are in use. Once they are removed from service, the monitor and any error messages often end up disassociated from each other, thereby upping the level of difficulty to mitigate or even identify any issues.
One of the newer and more difficult challenges with regard to portable and body-worn monitors is the fact that such devices are being sealed to keep moisture out and make cleaning and decontaminating easy, without the possibility of damaging the electronics. Adding connectors or attempting to combine functions on connectors increases bulk, expense or system complexity at the sensor ports.
Reading and Writing Relevant Data
Having a single readable, reliable traceable data source from manufacturing through operational status is a valuable tool for keeping these assets in service. Let’s take a look at some examples, starting with the manufacturing process. The manufacturing date, revision level, production line and location, serial number and a myriad of other types of data have long been jotted on sticky labels, using lot codes and other condensed methods. This type of information is basic for quality control and equipment traceability.
Today’s system requires option configuration, multiple calibration constants for sensors, service interval data, and more. Some solutions have user-programmable hot keys that a care facility could utilize to set and lock these functions. So much for the housekeeping data—what about the “check engine light”? Recording and having access to real-time error events can drastically reduce maintenance and out-of-service times.
One means of enabling this is to provide each piece of monitoring equipment with an RFID tag that connects to it via an I²C port.
Dual-interface memory is versatile.
Depending on memory requirements, these dual-interface memory chips may be multi-banked using one compliant I²C bus, and share the same antenna. Not only does this solution expand the memory possibilities, but the designer can take advantage of the 32-bit password security in all or part of any of the devices, thus granting conditional access.
M24LR64 Dual-interface RFID EEPROM
By now, you are probably asking, What happens if the device receives system commands at the same time I want to read or write something via RFID? As most engineers know, designing a simple system usually moves the complexity into the device. STMicroelectronics‘ M24LR64-R dual-interface EEPROM memory chip, for example, has built-in circuitry able to handle possible concurrent communications and powering activities from the RF and I²C sides.
Design Standards for Monitoring Equipment
Design standards for patient-monitoring equipment are a complex matrix, with dependencies on which patients are being monitored, and where this occurs. As mentioned earlier, evolving technology and standards present issues requiring the close tracking of manufacturing and maintenance data. Another difficult area to deal with is counterfeit accessories, sensors and other patient-monitoring apparatus. For attachments that plug in directly, it is possible to include some method of encryption that can be read by the host, which of course applies only to smart sensors and the like. For disposable attachments, the designer might want to include a low-cost RFID reader capable of interrogating an RFID tag embedded in the mating fitting. A secure challenge code could then be programmed into the dual-interface RFID chip.
As new accessories become available, it is a simple task to add challenge codes to the monitor. Counterfeiting is a growing and serious issue that requires a low-cost and reliable solution such as this.
ISO, Interoperability and Safety
Current dual-interface RFID chips employ the ISO/IEC 18000-3 mode 1 air-interface protocol (based on the ISO 15693 standard) that operates in the 13.56 MHz band. This standard, which supports up to a 1-meter (3.3-foot) range, depending on antenna size and other factors, is currently in use throughout the world. What’s more, it operates at low levels of energy considered to be very safe, and has readers available in many configurations. Recently, we have seen interrogators entering the market in cell phones with software support for Android.
Designer challenges are not getting any easier. Fortunately, the options presently available offer a myriad of solutions, sometimes targeted at non-related industries. This type of system appears to be finding a growing number of applications in the medical market, as designers recognize the simplicity and capability to solve a series of troublesome details in one low-cost, low-power and easy-to-implement device.
Tom Lavallee is an STMicroelectronics senior principal engineer focused solely on the medical and health-care market in Southern California. He has been with ST for 26 years, holds a degree in electrical engineering and has worked in the company’s automotive, consumer, computer and strategic segments.