Part 1 of this special report on the use of RFID in the healthcare industry explained the Micro-Modular approach to using RFID in healthcare. In Part 2, the authors detail the processes involved in the Micro-Modular approach and how they can create benefits for healthcare providers. In this third and final part, the authors continue to make their case for how their “micro-modular” approach can deliver a healthy return on investment.
The Smallest of Small
There are a few companies working on developing RFID technology to electronically track and scan for gauze pads, sponges and bandages in the operating room. We believe in the near future we could eventually justify the expense of using RFID for supplies costing less $2 each, given the fact that RFID hardware will soon be a lower-priced commodity. There is currently an interim solution where healthcare providers can experience many of the advantages
|
|
of Micro-Modularity while planning for the eventual RF implementation. That solution is Asset Offloading, which will be tackled in Part 3 of this special report, next week.
Asset Off-loading
“Asset Off-loading” is a viable solution for noncore functions at a hospital. This existing closed-loop service program allows the healthcare provider to keep minimal stock on hand and greatly simplifying its surgical supply chain process. Additional benefits of a closed-loop distributor may be the off-loading of accountability to the product’s genealogy ownership and the cleaning and recycling of instruments and supplies. This closed-loop system provides an interim solution, and many of the benefits (to a lesser degree) realized on ROI of a full RFID implementation, thus making the investment in such a service cost-justifiable today. The only process required on the part of the healthcare organization is to provide the supply vendor a daily schedule of the procedures to be performed one day in advance.
When healthcare organizations are internally ready to implement RFID, they can integrate the supply chain software used by the asset off-loading supplier or a similar software product. There are several asset-tracking software packages specifically for healthcare organizations. However, due to the difficulty of proving ROI on low-cost asset items, these currently do not address all aspects of medical supplies in healthcare environment.
Field Proof: SRI/Surgical Express Model
SRI/Surgical Express has 10 national distribution centers to supply hospitals with their daily procedural supply needs. They are one of the most complete models of asset off-loading, with a full integration of RFID technology into Class II Medical Devices, and among the largest users to date of “multi-read” applications of RFID technology.
“The incorporation of multi-read RFID tags into over 1 million of our reusable surgical gowns and drapes eliminates the use of labor-intensive bar code scanning to track product usage. This technology offers improved inventory control and monitoring of product quality. Once the used products are returned to our processing facility, they are sorted, cleaned, inspected, packaged, sterilized and shipped back to the health care provider for use,” says SRI/Surgical Express CFO Charles Pope. “This closed-loop system eliminates the need for health care providers to stock on-hand inventory and greatly simplifies their surgical supply chain process.”
Each procedures set up, requires planning, time and materials. SRI/ Surgical Express uses an assembly line process with tags on the items and carts. The medical supply kits are assembled on a reader table and verified. The kits are then placed on a chart for that day’s scheduled procedures for that particular operating room. At the time of loading for delivery, the carts and supplies are again verified through another reader. At the end of the day, the carts and supply kits are returned, verified, sorted, cleaned, inspected, packaged, sterilized and shipped back to the healthcare provider for use.
Once a hospital, division or a department has been set up with asset off-loading, the following benefits are realized:
Inventory Control
• Elimination of expired items
• Reduced supply levels
• Improved supply quality
• Stock shortage alerts
• Faster inventory turns
• Reduced loss and theft of supplies
Captured Costs
• Capitalized expense moved to an operational expense; tax incentive for a “for-profit organization”
• Reduced investment in unused inventory
• Accurate and Itemized billing from vendor
• Increased order management
• Increase reimbursement
• Increased labor efficiencies
• Asset visibility
• Improved sequence counts
Hazardous Waste Reduction
• Verified sterilization process
• Vendor uses best-quality supplies
Internally, SRI/Surgical Express experienced an immediate reduction of labor costs by 5 percent. This cost reduction resulted from increased efficiencies in sorting the inbound material and asset visibility of medical items. These are the same processes followed by a central distribution materials management department in a hospital.Asset Off-loading Software
The following software offerings attempt to address devices, Instruments and supplies from a closed-loop supply chain standpoint. We researched four companies of which two responded.
GE Medical Systems Healthcare Services: Together AssetPlus and IntelliMotion provide total life-cycle management capabilities, maintenance management for clinical and non-clinical equipment, and equipment tracking throughout the healthcare enterprise. IntelliMotion uses radio frequency to precisely monitor and locate assets throughout the facility in order to improve productivity, reduce search times, increase department volumes and lower costs. This software includes site analysis and process-improvement services to help implement change and drive return on investment.
Honeywell Asset Locator adds to Honeywell Enterprise Buildings Integrator (EBI) system, providing single-point access to asset management, security, and facility management through your desktop monitor, giving the exact location of essential equipment and people within a building—on a floor, or in a specific room. Honeywell Asset Locator updates the position of tagged assets every four seconds, providing an up-to-the-minute record for use in building and personnel security, utilization trending, budget allocation, vendor management and regulatory compliance.
The Total Solution
There are several companies who have a suite of enterprise-based software packages that address the hospital’s supply chain. Each vendor has an “event management and global product” component available for the surgical department. These packages have three integrated components to their software suite: a financial package, a materials supply manager and a department specific package. We have been talking about the OR and the “peri operational” software, which is software specific to the surgical department. However, these packages currently don’t have an RFID integration component. The companies we contacted are all working on an RFID integration component that will be released with their future software suite packages. Below is a summary of the three companies that responded to our inquiries. The table below summarizes these companies and their software.
|
table 3:
Asset Off-loading Total Solution offerings |
|
|
Horizon Surgical Manager
|
|
|
Pathways Materials Management
|
|
|
Horizon Supply Source
|
|
|
Core Business Software: Healthcare Supply Chain Management
|
|
|
Surgical Instrument Management
|
|
|
CareSuite
|
|
Meeting in the Middle: An Approach to ROI
We contend that process of building the supply chain channel from the manufacturer to the distributor and from the healthcare provider to the distributor—working both ends toward the middle—logically addresses the total supply chain integration barriers of standards and gives the industry time to develop a robust infrastructure. This approach results from the study of how regulations, compliance, product tracking, and medical life cycle management issues—operational, administrative and financial—affect each supply chain component. This approach will also give the needed time for the respective standards, regulations and technology to develop a full supply chain solution.
Once the single entity (healthcare provider) has resolved the challenges within their four modular walls, an effective ROI will result. The entity (healthcare provider) can now extend the RFID implementation strategy to the distributor level. At the same time, the manufacturer will be working on achieving an ROI within its modular four walls. Therefore, the approach should resemble an “ends toward the middle” strategy, thus yielding a better overall ROI for the entire supply chain.
The primary reason we suggest a meeting in the middle, is due to the numerous complexities of regulations and the varying standards that apply when we reach the provider of products and services at the end of the chain. Standards could develop where the other individual supply chain links need not comply with a set of regulations and/ or standards: The most reasonable approach would be to develop a vertical industry standard within the existing EPC defined standards. These vertical industry standards are confined to a particular industry and they do not affect other industries’ supply chain. Below are some examples of standards that may affect different parts of the healthcare supply chain.
Healthcare Supply Chain Reporting and Standards
The healthcare industry requires, for the most part, standard uniformity of reporting (HIPAA, Sarbanes-Oxley Act, OSHA), tracking and safety approval (FDA, 21 CFR Part 11 compliance) and labeling compliance (HIBC, HIDA, GMP and ISO 9000 requirements).
Pending Regulations
There are two pending regulations. One is called “Restrictions on the Use of Certain Hazardous Substances in Electrical and Electronic Equipment” (RoHS). As of July 1, 2006, the regulation will prohibit the sale of electronics-based products that contain more than 0.01% of specific hazardous materials. Since traces of hazardous materials are found in many medical devices, this law will fundamentally change the way such products are created, from initial design to sourcing of core components. The second regulation, “Waste Electrical and Electronic Equipment” (WEEE), is intended to ensure that electronic products are recycled or disposed of properly.
Geographic Standards
Split standards are seen when some regulations affect the supply chain in only one geographic area (Florida State Pedigree laws pending January 2006).
Healthcare Communications Standards
Electromagnetic emission/interference EMC guidelines (TIR 18 Medical Devices, ANSI c63.18)
Wireless transmissions for wavelength transmissions (ISO/IEC 18000-3 Part 3 – Parameters for Air Interface Communications at 13.56 MHz)
Environmental and Safety Standards
Environmental and operational safety patient standards may be relevant only to the healthcare provider: Joint Commission on the Accreditation of Healthcare Organizations (JCAHO) standards are directly related to safety, addressing such issues as medication use, infection control, surgery and anesthesia, transfusions, restraint and seclusion, staffing and staff competence, fire safety, medical equipment, emergency management and security.
EPC IS and Data Visibility
The EPCglobal Network promises a lot to manufacturers, retailers, healthcare providers, hospitals and many more industries in the ecosystem. The EPCglobal Network brings visibility of assets, reduction in inventories, just-in-time inventory handling, reduction in labor and many other benefits.
While we appreciate the benefits of RFID and the EPCglobal Network, the amount of data that flows on the network and the overhead for maintaining the data becomes an obvious concern. Also, when a particular object moves from one level to the other in the supply chain, more information about the object would be added for its usage at the next level.
If your business was looking for efficient systems that are relatively cheaper, help you in reducing maintenance and reducing the overhead, and at the same time provide security, you need to attain a low cost of ownership.
Defining Low Cost of Ownership
The authors define Low cost of ownership as sharing of information common to organizations that would ensure better security and data exchange mechanisms. This low cost of ownership will result into smoother operations and eliminating the storage of repetitive data. This means information about any object that is common should be kept at one place rather than at each level in the supply chain storing the same information. The storage of repetitive data increases overhead. If an organization were given a choice of storing less data and, at the same time getting all the information they need through other systems, it will solve a lot of overhead problems. “Low cost of ownership has to do with improving process to remove waste, improve accuracy of inventory, by improving information quality”, says Steve Heap of RFID Exchange, an RFID consultancy firm based in Northern Virginia.Attaining Low Cost of Ownership
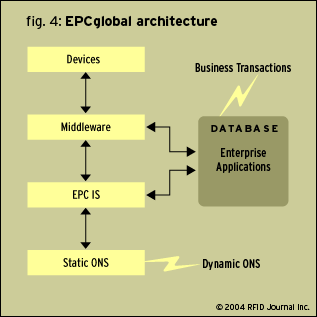
Figure 4 shows an overview of the EPC Global proposed architecture and various stages of interaction with other systems. One of the major challenges faced on the ONS and EPC IS specifications is security. Security becomes a major concern when unknown vendors and competitors look at information that can expose your company. With the EPC network in place, the information about the product will be on a network that can be accessible by unauthorized vendors. This can become a threat if competitors get information about your products at every level in the supply chain. Currently EPC Global is addressing this issue by taking various parameters into consideration.
Dynamic ONS defined in Figure 4 will give a list of the pointers (locations) where EPC (product) has been to date. Information about each product can be obtained from the product’s manufacturer’s database. Once the product moves to the next level in the supply chain—for example, from the manufacturer to wholesaler—and if there is more information added by the wholesaler at the next level, there will be one more EPC IS at the wholesaler level. The information about the product can then be obtained from the supplier as well as the manufacturer. If proper security aspects are not implemented, competitors could have total access (in a total trusting environment) concerning any product and they could gain an unfair competitive advantage.
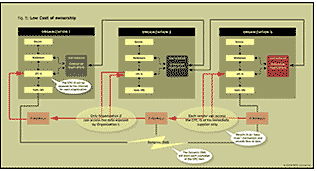
As explained in the Figure 5 below, the authors envisage the involvement of multiple vendors having their enterprise systems RF-enabled. The authors here try to abide by the definition of LCO (low cost of ownership) giving security and an “easy trust” mechanism for vendors at each level.
Low cost of ownership: Postulate
Figure 5 depicts that a vendor at any level can access only the EPC-IS of his direct supplier. The existing model suggests the data from the manufacturers EPC-IS is exposed to others in the supply chain. With the ability to only link to one’s direct supplier. This security limitation will prevent a lot of negative impact on the usage of RFID enabled systems on the network.
Figure 5 shows three organizations in the supply chain. The flow of the product from Organization1 to Organization 3 is through Organization 2. At each stage, the owner of the product and the products information is updated in the ONS (a pointer to the information about the what and where the product is), and each organization has its own EPC IS for exposing a variety of information pertaining to the item. Users can get the information in the Physical Markup Language format by querying the EPC IS. Further, if Organization 3 requires any information pertaining to this item, it will need only to contact Organization 2. If Organization 2 has that information, it will pass it along or else it will contact Organization 1. This ensures security of Organization 1’s product information so that Organization 1 does not have to worry about who is accessing its EPC IS and about what information is being sent to a party that Organization 1 doesn’t know, as long as there is an understanding trust agreement between Organization 1 and Organization 2. When the authors were discussing this scenario, optimization of information flow and the degree of trust as well as accountability to sharing that information were issues that were a major concern in developing this model.
We feel we have offered one viable solution to these trust and accountability problems. It was also noted that with the increase in the number of lookups, EPC IS performance might get affected. To get a work-around, one would need to implement caching of data in an efficient manner.
Another area where the ownership cost can be reduced will be to use systems that are globally accepted. This includes the systems that are based on open standards and are open-sourced. Using open standards and open source will reduce the risk as well as Integration costs when multiple systems from multiple suppliers are involved, as we see in the next section.
Standards and Source: Open and Governed
Open standards begin when a there is a unified collaboration of interested parties. This collaboration results in agreeing on specifications for implementing common requirements, i.e., open standards. This has happened at the Auto-ID Center and its successor, EPCglobal, which has promoted the development of open standards concerning automatic identification using radio frequency within the supply chain. These standards involve the reader protocol, EPS IS, ONS and PML standards that define the communication between entities of the supply chain. Using open standards endorses a commonly understood language across supply chain implementations of RFID. The choice to use open standards over the alternatives like proprietary standards and systems can improve one’s ability to realize common objectives.Physical Markup Language: Projected Evolution
Physical Markup Language (PML), for example, is a standard that helps in defining properties of the object. For the consumption of products and services and the creation of internal solutions, using open standards can decrease your costs, speed time-to-market, expand available options for communicating with various systems and utilize resources by using existing and available applications based on open standards. Using open standards in the products services and internal applications you produce, will increase adoption, market acceptance of solutions and lower market barriers through decreased customer risk. An open standard also allows you to participate in faster growing markets. Combining every aspect covered in this paper, one can achieve more efficient and cost effective results by deploying RFID at a micro-modular level and then uses the “meeting in the middle” approach through open standards.
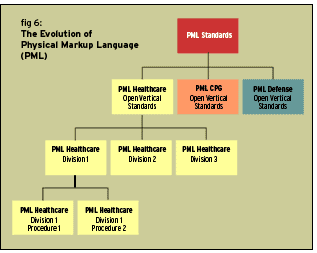
The adoption of Physical mark up language may take a while before it is accepted world wide as a commonly agreed upon standard language. Figure 6 in this section explains the authors’ views on the PML Standards and its evolution for vertical industry standards and divisions within that vertical industry i.e. P1 (P, Procedure) associated within that division for the healthcare industry. Vertical Industries will have to get together to agree upon a common format of language or structure or both. However, given the implementation of suggested vertical industry standard approach within the EPC standards through the extended PML constructs, seamless computing with interactions between industries could become easier. In fact, RFID merges the IT Industry with all the other functions within industries and across all industries.
Productivity will increase industry interactions and expand business to other vertical industries with these governed standards in place. Open standards will yield a better return on investment, as vendors will not have to comply with proprietary standards for communication.
As explained in Figure 6, these OVSs (open vertical standards) will attach to the main PML standard to define the vertical industry. The Industry OVS can have an attachment to its standard for a division or an open vertical standard division (PML.OVS.DIVISION1). The division OVSD than can have an attachment to it’s standard for the end product or service or an open vertical standard division product (PML.OVS.DIVISION1.PRODUCT1). All these attachments will tie back to the EPCglobal PML standards.
This means that there will be relatively low investments because multiple providers are brought into the picture. There would not be a need to invest more resources in terms of time and money to comply with proprietary standards. The systems would work with any vendor, without any changes, as long as they are complying with the open standards.
A Hardware Resolution
“We believe another part of the solution to open vertical standards is found in the hardware construction of the EPC tag. In a discussion with one of our reviewers, revealed that using a tag having 256 bits, of which 128bits are dedicated to the EPC Code, there would be an additional 128bits remaining to define industry, division and product specifications within that industry.” —Liz Churchill, Director of Life Sciences Solutions for RFID systems provider Matrics
In a Perfect World
A perfect world would have an open source code so that there would be no proprietary capturing of knowledge. Great progress would be made by having the same common language and standards so all companies could develop their best product knowing that it would work with any other product. Crossbow Technology, a provider of sensor network technology, is one company trying to make this world a reality. One of Crossbow’s main supporters is Intel.
Crossbow has its programs in Tiny OS, an open source extendable and scalable platform. It is in the process of developing an out-of-the-box solution that is a customizable interface (GUI) middleware application on its tiny RFID readers, which the company calls motes. A single board Linux computer, known as the “Stargate,” powers the whole system. Sam Godwin, Crossbow’s VP of sales and marketing, believes that the mote (reader) will be the future middleware of the RFID system, interfacing to the backbone of the ERP application software.
Sam Godwin predicts that within two years these intelligent motes (readers) will sell for about $10 each.Conclusion
We have given suggestions on what needs to be considered, why the Micro-Modularity approach is valid, how to overcome the security and data-transfer issues (See “EPC IS and Data Visibility: Projecting a Low Cost of Ownership”) and where we can address the problems (see “Physical Markup Language: Its Projected Evolution,” above) There are only two questions left for healthcare to implement RFID, and one of those questions is, When it will happen? The when and why will drive the future of this industry. Both when and why are intertwined with the client’s needs. Often the client knows it can improve its management of the supply chain. However in a new industry, there are numerous options with many valid approaches. The importance is to provide a clear path for the client.
We believe we have shown that by the tagging of limited value assets ($90-$5,000) there are additional gains to an organization’s labor efficiency, inventory management, risk management (patient safety), and resource standardization and utilization, that can not be accounted for by tagging only high asset value items.
Inevitably, it comes down to timing. Do we know the industry regulations and how they impact our client? Have we asked the right questions to understand the client’s problem? Does the client believe that by addressing their internal operation, there will be greater returns than if they where to concentrate on another area of there operation? Finally, have we approached enough potential clients to gain momentum within their industry?
We wrote this study because as long as there is uncertainty in a market, people will be hesitant to invest. We took a risk as independent authors to solidify our industry’s direction in demonstrating how a vertical system approach to an industry will help RFID gain faster acceptance in every industry. We offered our future clients tangible proof that there are ways to look at your assets “true value” to find an operations hidden efficiency’s. Finally, we determined the minimum breakeven point for investment into an RFID initiative.
Contacts and references
|
Company/Person |
Web Address/Email Contact |
|
Patni |
|
|
Mobile Aspects |
|
|
SRI/ Surgical Express |
|
|
Crossbow Technology Inc. |
|
|
Honeywell |
|
|
GE Healthcare Asset tracking |
|
|
McKesson Corporation |
|
|
Lawson Software |
|
|
Picis |
|
|
Oat Systems |
|
|
Siemens Medical |
|
|
Agility Healthcare solutions |
|
|
Precision Dynamics Corporation |
|
|
Texas Instruments (RFID div.) |
|
|
Matrics |
|
|
Digi International |
|
|
EM Microelectronic-Marin SA. |
|
|
RF Code |
|
|
AI Technology |
|
|
InfoWave Solutions |
|
|
ODIN Technologies |
|
|
Association of OR Nurses |
|
|
RfidEXchange |
|
|
Accenture |
|
|
Advanced Medical Technology Assoc. |
|
|
Global Medical Device Nomenclature |
|
|
Susquehanna Health System |
|
|
Ingalls Same Day Surgery |
|
|
Ingalls Hospital |
|
|
The Midwest Sinus Center |
|
|
Rush Hospital |
|
|
Yeatman and Sons |
|
|
Ken R. Thompson Consulting |
|
|
Joe Rodgers |