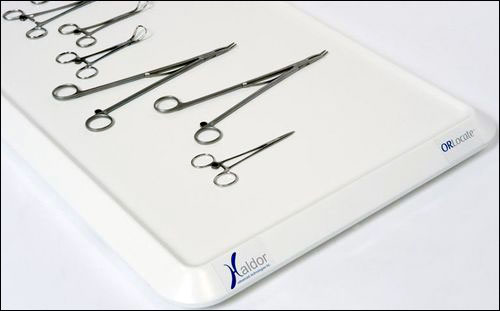
Having received U.S. Food and Drug Administration (FDA) clearance this month for its surgical sponge and instrument tracking technology, Haldor Advanced Technologies plans to begin trialing its ORLocate system at three U.S. academic hospitals in October. The system, whereby surgical sponges and instruments are fitted with passive high-frequency (HF) RFID tags, has been previously tested in a lab environment and with animals. The company believes ORLocate could help hospitals ensure that equipment and materials are not misplaced during surgery, either in a patient or in waste containers, and track the use, maintenance and status of instruments.
According to Jacob Poremba, the cofounder, president and CEO of Haldor USA (an American subsidiary of the Israeli firm), the solution not only promises to increase safety in the surgical room, it also reduces the chances of shrinkage—by identifying when an instrument is missing from its tray, and by enabling an RFID-based search for that item. The system also saves staff members time otherwise spent in the arduous process of counting instruments and sponges prior to surgery, and again after a patient is closed up.
To obtain the FDA’s permission to market the product to U.S. health-care providers, Haldor needed to show that the technology was safe to use in a surgical setting. As part of that process, the company submitted the results of biocompatibility testing (which looks for toxicity or other hazards to patients that might result from the technology’s use). Those tests were carried out by NAMSA, an independent medical device contract research organization located in Northwood, Ohio, as well as by labs in Germany and Israel.
The complete system consists of multiple components, including an ORLocate console with a 19-inch touch screen, on which information regarding the items present, along with their status, is displayed by the system’s Sterile Processing and Distribution (SPD) software. In addition, there is a sponge tray with an RFID antenna and a reader for sterilized sponges, as well as a sponge bucket, also with a reader and an antenna, used to count soiled sponges. For hemostats and other surgical instruments fitted with tags, there is an RFID-enabled “back tray,” located on the surgical setup table on which the hospital’s trays of instruments are stored in preparation for use, as well as an RFID-enabled Mayo tray, used to detect instruments placed on the Mayo stand as they await surgical use. A handheld reader, known as a locator, is utilized to pinpoint missing items, and a desktop reader dedicated to the sterilization procedure captures each tag’s ID number as instruments are sterilized following a surgical procedure. Finally, there are RFID antennas located on the console’s upper panel and near the Mayo tray, to identify when new sponges or tools are added during surgery.
The ORLocate system includes RFID tags for instruments. Each tag, explains Donald Mudd, the general manager of Surgical Investments LLC (ORLocate’s exclusive U.S. distributor), can sustain at least 1,000 sterilization procedures, as well RFID-enabled sponges or tags to apply to existing sponges. The tags, the company reports, have proven to read well in environments containing large quantities of metal and liquids, and can also sustain the sterilization process thanks to the encapsulation of the tags that Haldor’s engineers have developed.
Each tag is encoded with a unique ID number and operates at 13.56 MHz, in accordance with the ISO 15693 RFID standard. The instrument tags can be applied to any flat surface measuring 3 millimeters (0.12 inch) or larger. The SPD software can stand alone on a hospital’s server, or be integrated into a facility’s existing back-end system.
The system, according to the company, offers a solution to the existing time-consuming practice of manually counting sponges and instruments before and after a surgical procedure to ensure nothing was left within a patient. That manual process is usually completed by two nurses for each procedure. Even with that process in place, items are still occasionally missed—and if staff members determine that an asset is missing during surgery, they must visually search for that item before the surgery is completed. “Not only is that a laborious process, it can be very costly,” Mudd says. On average, he points out, “it costs $150 to $400 per minute to run an OR.” By allowing employees to use a handheld reader to locate the missing item, valuable time can be saved.
Initially, ORLocate’s staff will attach the RFID tags—which it manufactures itself—to the instruments and sponges, and then enter data about each item into the SPD software, such as the instrument’s size and style, thereby providing a record of that unique item. Each tray on which items are stored is also fitted with an RFID tag, the unique ID number of which is married to the tag IDs of those instruments or sponges.
When surgery is scheduled for the next day, hospital workers pull the necessary trays of surgical equipment, each of which could contain hundreds of items. Although smaller assets are not tagged, they are listed in the SPD software system and are linked to each tray, so that workers can view a display of what should be present within that particular tray.
As the trays and the instruments within them are placed on the ORLocate back and Mayo trays, the corresponding reader captures each instrument’s unique ID number and forwards that information to the SPD software via a cabled connection. The software then displays each tool’s status, indicating whether that item is on the back tray or on the Mayo tray. ORLocate also tracks tagged sponges as they are placed on the sterilized sponge tray. As each tagged item (either an instrument or a sponge) is removed for use in the procedure, the reader ceases to read that ID—a tag needs to be within 45 centimeters (17.7 inches) of a tray in order to be read—and the software is updated to indicate that the item is in use. Once an instrument is utilized, it is returned to the tray, and its status is then updated. A soiled sponge is placed in the sponge bucket, which also reads that sponge’s ID number and updates its status as having been used. All items too small to be tagged must be manually counted before the operation begins, and again after it ends. The nurse manually inputs the initial count of untagged item into the SPD software, prior to the start of surgery.
At the end of the surgical procedure, the nurse pushes the “end surgery” tab on ORLocate’s SDP software, and the software then determines if all tagged items are accounted for, and also reminds the nurse to manually count all untagged items. If a tagged item is unaccounted for, an alert is flashed on the screen and a warning sound is emitted. If unable to locate a tagged item, the staff can then utilize a handheld interrogator with a read range of approximately 22 inches, by waving it over the patient or waste containers.
Once a surgical procedure is completed, the software closes that surgical record as properly completed, though it will not close if a tagged instrument or sponge is missing, unless the software is forced to close by an authorized individual. Everything is recorded in the database and printed out in a report for the hospital staff following the procedure’s completion.
The software can issue an alert if a tagged instrument is found to be missing during sterilization, as well as store data indicating when each tool is sterilized. The software also tracks the number of uses and sterilizations each piece of tagged equipment has undergone, thereby allowing hospital management to know when a piece has been used its maximum number of times, or is due for maintenance, such as sharpening.
If a physician finds that a tagged instrument does not operate satisfactorily during surgery, a module in the software allows hospital employees to read that instrument’s tag with the handheld reader, and input any problems it is experiencing. The software will then not allow the tool to be reused until the software has been updated to indicate that the item has been properly repaired by an authorized individual.
The technology list price is slightly less than $100,000 per surgical room, Mudd says, including the console, RFID readers, RFID-enabled back and Mayo trays, SPD software and 200 tags. However, he notes, the price per surgical room could be significantly less for high-volume purchases. The system is designed not only to ensure patient safety and reduce manual counts—which are time-consuming and error-prone—but also to reduce shrinkage by alerting workers whenever an item fails to be returned to a tray during surgery or the sterilization process. (In some cases during the sterilization procedure, hospitals experience a loss of small surgical equipment, which can become missing at some point after a procedure.)
ORLocate is not the first system on the market that uses radio frequency identification to track sponges. In 2007, for example, ClearCount Medical Solutions, a Pittsburgh-based company focused on improving surgical safety, received FDA clearance for its SmartSponge System (see RFID-enabled Surgical Sponges a Step Closer to OR). Although there are currently other RFID solutions in place for tracking sponges, Mudd says, “there is no other company that (is tagging) sponges and instruments.”