With the number of RFID-related patents and litigations on the rise, the pharma industry should give more consideration to evaluating RFID patent portfolios when selecting RFID vendors to implement RFID drug pedigree programs.
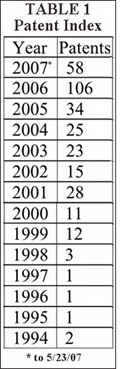
Over the past two years, RFID-related technology and patents in the United States have blossomed, and the volume of patents issued annually in RFID technology reflects this technology growth. Tracking the number of patents with the term “RFID” in their titles and issued by the U.S. Patent and Trademark Office (USPTO) between 1994 and 2007, provides a quick and easily accessible index of the growth in the issuance of patents related to RFID technology (see Table 1, “Patent Index,” below left).
The U.S. Patent and Trademark Office estimates that approximately 6,000 patents related to RFID technology have been issued in the United States. A corresponding increase in pending patent applications often accompanies an increase in issued patents, and USPTO estimates there are also approximately 6,000 RFID-related pending patent applications as well.
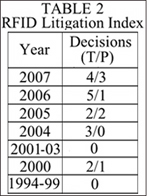
With this burgeoning of RFID technology and patents, has come increased litigation in the United States in this field, with a significant portion of the federal cases in this area related to patent issues. Tracking the number of reported decisions using the search term “RFID” in a LEXIS search of reported federal court decisions provides a ready index of the growth of RFID-related patent litigation.
The total RFID-related federal decisions from this search (T) is reported under “Decisions” in Table 2, “RFID Litigation Index” (below right), followed by the number of decisions involving patents (P). Seven of the 16 reported RFID-related federal court decisions in the past 13 years have involved patent litigation.
A patent holder may sue any infringer of its patent, including the customer of a competing manufacturer. Thus, as major customers of RFID technology, pharma companies have an exposure to patent-infringement litigation and can be caught in a squeeze play between competing RFID vendors.
Concomitant with the increase in RFID patent filings and RFID-related litigation has been a demand in the pharma industry for reliable electronic track-and-trace technology to help secure the integrity of the pharmaceutical supply chain by providing an accurate drug pedigree.
On June 8, 2006, the U.S. Food and Drug Administration (FDA) issued its most recent statement, the “FDA Counterfeit Drug Task Force Report: 2006 Update” (see FDA Issues New ‘Counterfeit Drug Task Force’ Report).
While the report recognized that the FDA’s earlier goal “that widespread implementation of e-pedigree” by 2007 would not be met, it added that the FDA “continue[s] to believe that RFID is the most promising technology for electronic track and trace across the drug supply chain.” In addition, the report described several obstacles to wider RFID adoption, including:
- A lack of standards (for e-pedigree fields and format, data systems, international transmission standards and hardware specifications).
- Privacy concerns.
- Concerns regarding the ownership of confidential business transaction data.
- Challenges involved in serializing all products.
- Concerns over the accuracy and speed of electronic devices and systems.
- A lack of definitive data to determine how RFID would affect sensitive products (for instance, liquids and biologics).
RFID companies have actively focused on overcoming these obstacles. In particular, among other pharma market concerns, they have sought patent protection on methods and technologies related to improving the accuracy and speed of RFID devices and systems, as well as protecting the security of the information in RFID tags.
The trends reflected in both tables, coupled with the demand in the pharma industry for improved RFID technology for use with drug pedigrees, suggests that to limit their exposure to possible patent-infringement litigation, pharma companies may wish to evaluate patents as part of their selection process when choosing RFID vendors. The possibility of patent litigation should be a significant concern to drug companies considering substantial investments in RFID technology, because there is the potential for a permanent injunction to be issued barring the use of the infringing technology.
A customer of RFID technology will, in its purchase agreements with its technology providers, receive indemnifications against patent-infringement liability. Still, prudent pharma companies should seek to limit their exposure, to avoid being caught up in patent litigation between competing RFID companies.
Marilyn Neiman is a senior counsel at Cohen Pontani Lieberman & Pavane LLP, a New York law firm focused on intellectual property litigation. Neiman is a litigator with more than 25 years of experience as a counsel in many patent, trademark and copyright cases throughout the United States, encompassing a wide variety of industries, including medical, jewelry and toys.