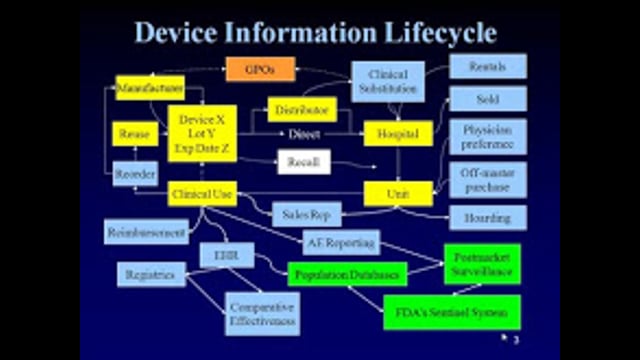
Tracking and tracing the path of medical devices from manufacture and distribution to the point of use and, ultimately, the end of the product’s life cycle—including possible recalls—has broad implications within today’s health-care community. The FDA’s Unique Device Identification (UDI) Rule will establish a single device-identification system that is consistent, unambiguous, standardized and globally harmonized. All medical device manufacturers will be required to comply with the new UDI methodology. In this session, learn how a UDI system will provide for the early detection of defective devices, as well as facilitate device recalls, in order to enhance patient safety and reduce medical errors.
Speaker:
Jay Crowley, Senior Advisor for Patient Safety, Center for Devices and Radiological Health , FDA