The U.S. Food and Drug Administration (FDA) averages between 350 and 375 medication recalls annually—a number that has recently been on the rise, according to Mike Molby, the director of pharmacy services at CaroMont Health‘s CaroMont Regional Medical Center, who receives and must address such recalls. The majority of these requests, he says—approximately 300 of them—affect the Gastonia, N.C. hospital. With each recall, Molby reports, the hospital’s pharmacy must determine if it uses that medicine, and then locate and remove the drug in order to ensure that it is not administered to a patient. In some cases, the medications are stored on crash carts distributed throughout the hospital. Pharmacy personnel must search for those carts, open each sealed kit and then return those kits to the pharmacy to be restocked, resealed and placed back on the cart.
The hospital has eliminated much of that manual process, however, thanks to the installation in May 2013 of a radio frequency identification-based solution provided by Kit Check. The solution consists of Zebra Technologies printers, passive ultrahigh-frequency (UHF) readers built into a Kit Check scanning station, and UHF EPC RFID tags attached to medications, as well as to the kits in which they are packed. The technology enables users to know if any kits contain recalled drugs, as well as which ones, simply by accessing the Kit Check software. Since the system’s installation, Molby reports, it has eliminated the need for workers to manually search each crash cart during a recall, while also ensuring that the identification of recalled medications proceded quickly.
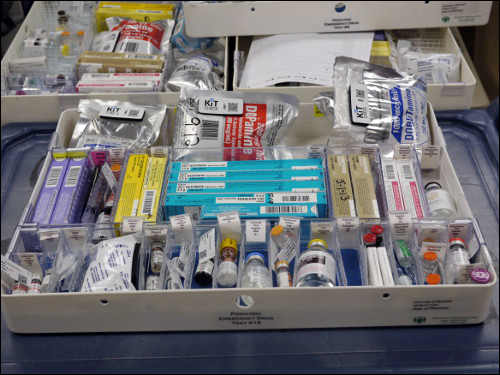
The CaroMont Regional Medical Center is a 435-bed hospital with 96 crash carts containing emergency medical kits, as well as several dozen independent kits at stationary locations, each filled with 20 to 50 medications or other emergency items required by patients in urgent need of treatment for a stroke or some other condition. What makes CaroMont unique, says Kevin MacDonald, Kit Check’s co-founder and CEO, is the speed at which the hospital deployed the system: The facility’s pharmacy had tagged the majority of medications used in the kits within only about two days.
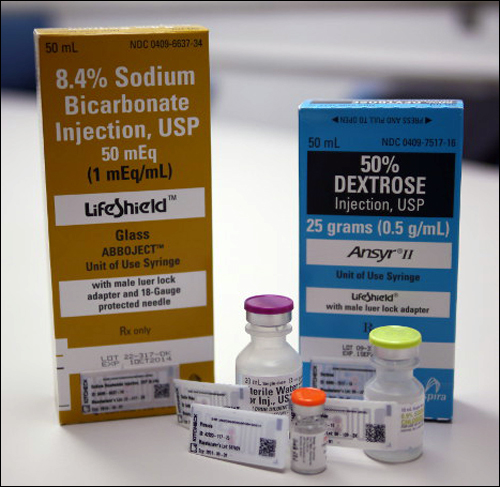
With the Kit Check technology, MacDonald explains, the hospital staff first scans bar codes on medications, and then prints an EPC UHF RFID tag on a Zebra R110 xi4 printer. The tag is applied to the medication, while the collected data is stored in the Kit Check software residing on the company’s hosted server, and is available to hospital personnel via a password. The printer then encodes and prints a tag for each kit, in order to identify its category—for example, a stroke treatment kit for an adult male patient.
Once medications are packed within the kit, it is placed inside the scanning station, which comes with a built-in RFID reader. According to MacDonald, the type of reader installed in the stations varies. The station’s reader interrogates all tags and forwards that data to the software via a wired connection to a PC. Based on the tag reads, the Kit Check software determines if the medications match that kit’s requirements. It then stores the results, displaying an alert if the kit is missing medications or contains an incorrect drug.
The kit-scanning process can be completed in approximately five minutes, the company reports, as opposed to the 20 minutes required to manually check that the proper pharmaceuticals are packed in the kit before it is sealed. Staff members then assign the kit to a cart and send it to the appropriate hospital department.
If a recall occurs, workers can enter the recalled medication’s lot number into the Kit Check software, which identifies if that drug is packed in any kits, pinpointing the affected kits and the carts on which they can be found. Employees then manually locate the appropriate carts and kits, returning them to the pharmacy and removing the recalled drug.
Prior to the Kit Check system’s installation, workers had to walk to every cart within each department, visually identify kits that might contain a recalled medication and break each seal to look inside. The kits then needed to be resealed, even if the recalled drug was not present. That task could often take several hours or days to complete, MacDonald says. But with the RFID solution in place, he notes, the response to a typical recall can be reduced from about 20 to 30 hours, down to only around two hours.
The technology also enables a more efficient process of replenishing kits after medications are used on a patient, the company claims. What’s more, it facilitates the identification and replacement of expiring drugs.
To date, the CaroMont Regional Medical Center has tagged about 15,000 medications with Kit Check UHF tags. The tags (the chip vendor of which Kit Check declines to specify) include the RFID company’s proprietary design to make it possible to read multiple tags simultaneously within a highly dense environment containing liquids or metal packaging. The system, MacDonald says, provides a 100 percent read rate for tags inside and on the kits.
“It’s fairly simple to use,” Molby states. Personnal had to be trained to apply tags and use the scanner, he notes, “but they get it, really quick. I think they see that we’re on the forefront of something pretty advanced.” Molby says he expects to achieve a return on investment within one year, based on reduced labor time.
The Kit Check solution is also being used by the University of Maryland Medical Center (UMMC), a Baltimore hospital approximately twice the size of the CaroMont facility (see Hospital Pharmacy Keeps Emergency Medication Kits in Check). In the case of UMMC, Avery Dennison inlays are being utilized with Impinj Monza RFID chips. UMMC did not respond to a request for an update regarding the technology’s use.