The Axia Institute’s RFID Lab worked with pharmaceutical supply chain constituents and technology providers to simulate the prescription drug supply chain to test RFID’s effectiveness.
 The Axia Institute at Michigan State University recently completed a 15-month, lab-based RFID project to track prescription drugs across the supply chain. The results, the researchers say, demonstrate that UHF RFID technology can reliably track products through distribution and shipping.
The Axia Institute at Michigan State University recently completed a 15-month, lab-based RFID project to track prescription drugs across the supply chain. The results, the researchers say, demonstrate that UHF RFID technology can reliably track products through distribution and shipping.
The testing results come just as the FDA’s Drug Supply Chain Security Act (DSCSA) requirements are enacted in late November. Due to concerns from some dispensers, the provision will not be officially enforced until November 2024.

Dr. Bahar Aliakbarian, Associate Professor, Department of Biosystems and Agricultural Engineering and Director of Research and Development at the Axia Institute at Michigan State University
The institute’s Axia Lab began the program of simulating a prescription drug supply chain – and testing RFID’s effectiveness in that environment – in 2022. The testing was completed this fall, says Bahar Aliakbarian, Axia’s senior R&D director. AXIA Lab will present the results of its demonstration and piloting in a webinar on Nov 13.
Axia Lab is a value chain research laboratory that examines RFID technology use in healthcare, food, and agriculture, as well as advanced manufacturing. The lab was first opened in 2020 and since then has conducted testing validation of tagged pharmaceuticals as well as consumer goods and other projects, explains John D. Hatfield, The Axia Institute’s executive director.
They targeted the prescription drug process to address questions expressed by pharmaceutical companies about the feasibility of using RFID to help with DSCSA requirements. While the DSCSA does not specify RFID technology use, the automatic data capture from RFID tags could provide the unique digital identity of each product while offering other potential benefits, such as automated data capture and supply chain management. Therefore, the pharmaceutical industry was interested in a feasibility study of UHF RFID tags and their effectiveness in the supply chain.
Building a Pilot Advisory Group

John D. Hatfield, The Axia Institute’s Executive Director
“We got together with several different companies from the healthcare and pharmaceutical industries across the value chain,” says Hatfield, resulting in a pilot advisory group representing more than a dozen companies such as drug manufacturers, RFID solution providers, or healthcare firms.
The advisory group met at the Axia Institute and in a pharmaceutical manufacturing site, distribution center, and logistics site, as well as a hospital. That effort led to a simulation built in the Axia Lab that mimicked the real-world process flow of drugs to retail locations. One goal was to identify some of the weaknesses or gaps “within each of these format stakeholders within the supply chain that RFID could bring some benefits to,” says Hatfield.
The pilot used RFID tags from three providers, readers from Zebra, and printers from both Zebra and Printonix as well as software developed by ACSIS and Axia to manage read data and readers. The serial shipping container code (SSCC) labeling format based on the GS1 standard protocol was used for all three tags. Two tags were pre-encoded by the tag manufacturers, while the lab used the GS1 encoder system to encode the third type of tag in their lab manually “so we were also able to compare the encoding systems,” she says.
Linking Totes of Drugs with Palletized Shipments
AmerisourceBergen provided totes in which tagged items were packed. The lab tagged three major pharmaceutical products (using tags from other manufacturers) — one a liquid in a glass vial and two solid products in bottles.
 Once tagged products were packed in totes, they were read with a Zebra Technologies and SLS T-Series tunnel reader, and each product tag was read along with a parent tag applied to the tote. The software then linked the tote tag with the products packed inside it.
Once tagged products were packed in totes, they were read with a Zebra Technologies and SLS T-Series tunnel reader, and each product tag was read along with a parent tag applied to the tote. The software then linked the tote tag with the products packed inside it.
In the second step of the simulated journey, totes were stacked on a pallet, which was shrink-wrapped. A panel reader at that site read the tag on the pallet as it revolved twice during shrink wrapping and generated a pallet tag.
In the last stage of the shipping preparation, the loaded totes, in shrink-wrapped pallets, were transported through dock door portals mimicking the exit of the facility, and a Zebra and SLS portal reader with antennas on each side captured the tag ID of the pallet, which then indicated that the products had been shipped.
The tags were read using two timelines: the tunnel reader read product tags for 1.5 seconds and 2.5 seconds. For the shrink-wrapping RFID tag read, they piloted a 15-second read time, the typical amount of time necessary to wrap at least two layers of shrink wrap on a pallet. The tag reads were each tested 100 times.
The team also studied two different formulations within the totes, says Aliakbarian. In one case, totes had more solid products than liquid products, and in the second case, totes held a majority of liquid products. The aim, she says, “was to see if there is any interference for the readability.” They found that the contents did not significantly impact tag read effectiveness. “Additionally, we also evaluated the highest number of products that we can put into these totes,” she says.
When it came to testing tags, the group chose labels that were the same size, and all had an external memory for data, such as the product’s expiration date and its batch and lot number.
The results affirmed the researchers’ premise that the tags could be read at a high rate in this environment. Some tags performed better than others, but all provided readability rates higher than 96.5 percent. The presence of liquids or other environmental factors did not notably affect the results, she says. “So the importance is the discovery that tag design plays a more important role for the readability than the tag population,” or the environment. “We were, in the beginning, thinking that liquids were going to give us less readability than the solids, but then we have figured out that tag design was one of the major important factors,” she says.
Some tags provided higher than 99 percent read rates regarding tagged products and totes. They achieved 100 percent readability at the pallet level no matter what type of tag was used, she adds.
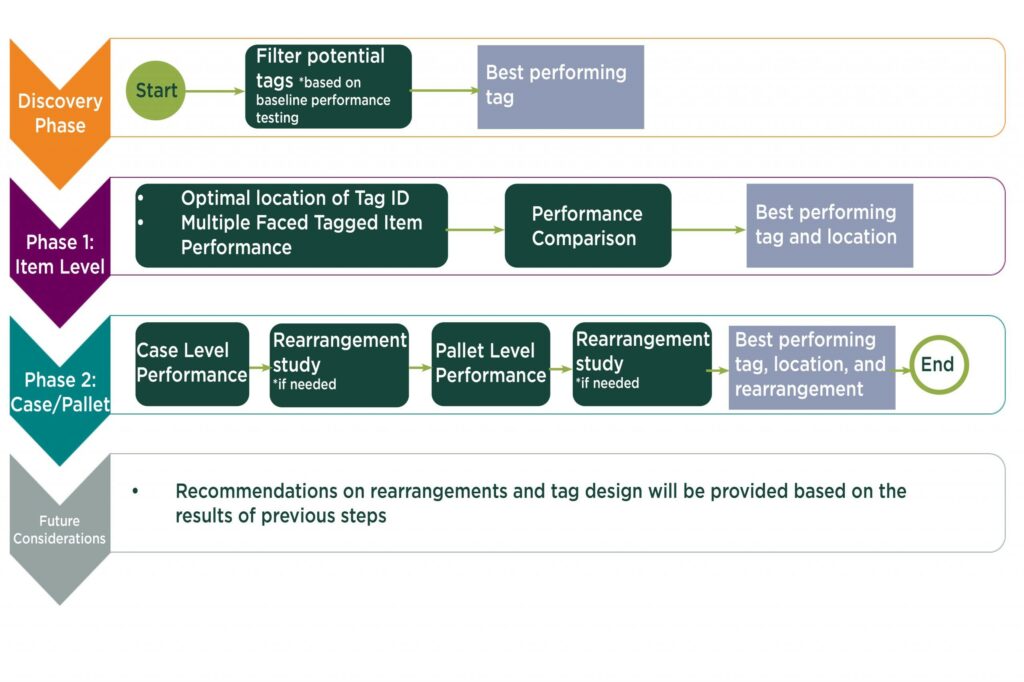
Axia’s process
With the pilot completed, Axia Lab is focused on sharing the results with the pharmaceutical and healthcare industry. “We want the industry to understand that RFID is viable for doing this sort of traceability work,” says Hatfield. In fact, the AXIA Institute composed comments about the viability of RFID to the FDA as the agency was accepting comments in later phases of the DSCSA. “While 2D barcodes are fine, ultimately RFID may be a good addition for the entire value chain for a number of reasons,” says Hatfield.
Now that the lab has confirmed the viability of RFID tag reads, the group hopes to work on other challenges or concerns, such as return on investment into the technology or comparison of the cost against reduction of errors or time savings. Pilot group members have indicated that the technology could help prevent mistakes in product identification in the supply chain by automating the data collection.
“We hope this is a tipping point to demonstrate that RFID technology can work in providing the important traceability of medications across the supply chain,” says Hatfield.
Another learning from the study, Hatfield expects, is a demonstration regarding how source tagging (by the drug manufacturer) could be helpful. If a product is tagged with RFID from the point of manufacture, it would reduce the need for tagging at the distribution center. At the same time, visibility of the product could be enabled from when the product leaves the production site, thereby offering end-to-end traceability.
Participants in the advisory group included ACSIS, Apotex, Cencora, CCL eAgile, Fed Ex, Fresenius Kabi, GS1, Sanofi Aventis, BarTender by Seagull Scientific, Zebra, and Texas Children’s Hospital, as well as Walgreens.
Key Takeaways:
- Axia Lab – a technology-neutral research group – has completed testing of RFID technology effectiveness in the drug supply chain.
- The results found that the type of tag impacts read rates, but all three tags tested provided 96.5 percent readability or higher.


