Apr 15, 2013When counterfeit drugs infiltrate a pharmaceutical supply chain, they can endanger the safety of patients and damage the reputations of legitimate manufacturers. For several years now, identifying pallets, cases and individual packages of medicine with unique Electronic Product Code numbers (via RFID or 2-D bar codes) and tracking them from manufacture through distribution and on to retail pharmacies has been promoted as a way to make the supply chain safer and more secure.
Several pilots have demonstrated that the hardware, software and network standards, such as EPC Information Services (EPCIS) for sharing serialized event data securely in near-real time, can verify chain of custody. The EPCIS standard provides a way to capture and share physical event data—each time an EPC tag is read as it moves through the supply chain, for example. Information regarding changes of containment, such as when items are packed into cases and bound to pallets, can also be recorded as EPCIS event data.

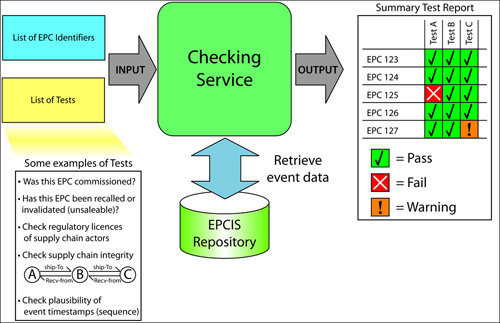
But one hurdle has remained. Given the large volumes of pharmaceutical packages in circulation, this solution would put a huge burden on companies that must check their shipments. In my previous column, A New Approach to Pharmaceutical E-Pedigrees, I introduced the concept of "Checking Services," a way to handle serialized data verification by automating the process or outsourcing it to an accredited checking operator.
Since October 2012, the Cambridge Auto-ID Lab has been actively working on the design of Checking Services within a new GS1 technical work group. We view Checking Services as a new component within the GS1 EPC network architecture. The goal is to standardize the interfaces, to ensure interoperability and develop robust accreditation requirements. That will let companies have a choice of providers and give them the confidence that the checks will be performed consistently and correctly.
We are just beginning to tackle the technical part of the standardizing work, and we expect more solution providers will become involved in the near future. Participation is important because technology providers will likely offer the service; just as there are multiple providers of EPCIS repositories, there will be many providers of Checking Services. Their prototypes must be well aligned with the direction of the standard and ready to meet expected legislation deadlines for electronic pedigrees.
Click here to view a larger version of the above table.
Checking Services will be able to automatically gather EPCIS event data, then perform a number of procedures on the data to identify any gaps or inconsistencies. Companies will be able to receive summary reports of the results before they receive the physical goods, enabling them to decide which to accept and which to quarantine for further investigation, without slowing the receiving process.
This work is initially intended to support the U.S. pharmaceutical sector, which will likely need to comply with e-pedigree legislation in 2015. But Checking Services will provide a flexible framework to allow multiple checks to be selected and configured, including those which might be defined in the future in response to traceability legislation in other sectors or regions.
Mark Harrison is director of the Cambridge Auto-ID Lab.
